The Government has formally issued guidance that children aged 5 to 11 can now receive COVID vaccinations as recommended by…
Continue Reading
On the day appointments available
Coronaviruses are a large family of viruses that cause respiratory infections ranging from the common cold to more serious diseases. COVID-19 is a disease caused by a new coronavirus form, first reported in December 2019 in Wuhan City in China. Symptoms of COVID-19 are variable, but often include headache, fever, loss of appetite, fatigue, nausea and mild to moderate respiratory illness such as coughing, sore throat and shortness of breath.
The COVID-19 virus spreads primarily from person to person through close contact with an infectious person, contact with aerosols from an infected person’s cough or sneeze or touching objects or surfaces that have aerosols from an infected person, and then touching your mouth or face. People who are 70 years or older and those with underlying medical conditions such as diabetes, chronic respiratory disease, and cancer are at higher risk of developing a serious illness. Since COVID-19 is a new disease, there is no existing immunity available in our community resulting in the spread of the virus quickly and widely.
The best way to protect your loved ones and yourself
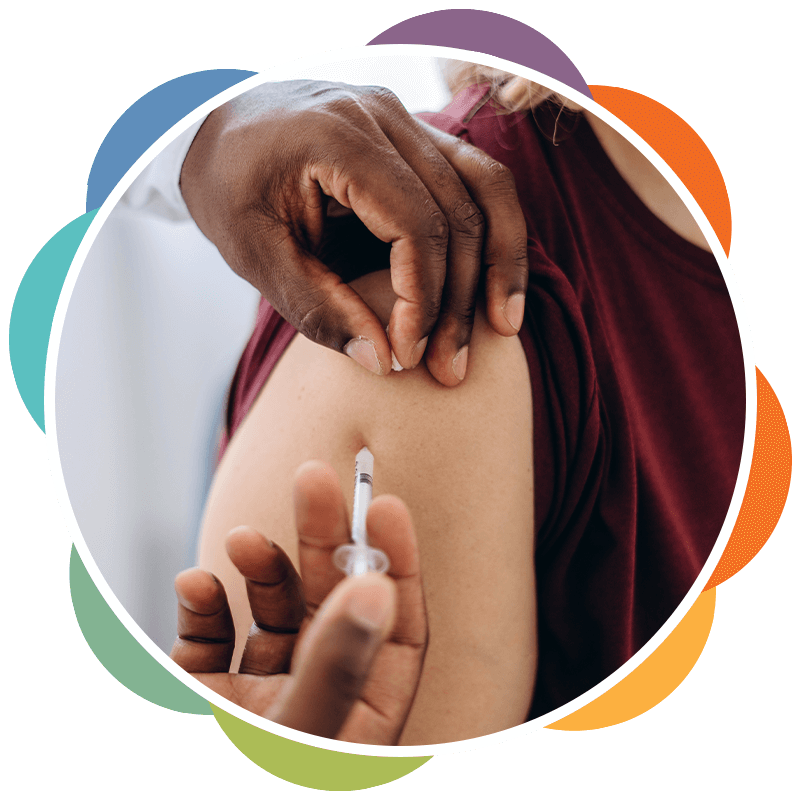
The best way to protect your loved ones and yourself is to get vaccinated for the COVID-19 virus. The Australian government has commenced the vaccination program in Victoria on 22nd February to provide free covid vaccines for all Victorians.
Due to the limited availability of covid vaccine in Victoria, initial vaccination will be offered using a “priority framework” where individuals who are at high risk of contracting COVID-19 such as quarantine and border workers, frontline health care workers and aged care and disability care staff workers along with elders, vulnerable patients and aged care and disability residents will be offered the vaccine first as the priority.
While COVID-19 vaccination is not mandatory, it would be better to vaccinate since COVID-19 can cause severe lung and generalised disease which could easily spread from one person to another with few or no symptoms and increase the risk of long-term effects on health, particularly in elders. By choosing to vaccinate, you are protecting yourself and others in the community from this deadly virus.
The Government has formally issued guidance that children aged 5 to 11 can now receive COVID vaccinations as recommended by…
Continue ReadingThe Government has formally issued guidance that children aged 5 to 11 can now receive COVID vaccinations as recommended by…
Continue Reading
To determine which vaccination is appropriate for you please visit the government eligibility checker or book a brief phone appointment with your GP here.
Both the Pfizer (Comirnaty) and AstraZeneca (Vaxzevria) vaccines are very effective at stopping people from becoming very sick if they catch COVID-19.
The vaccines have been thoroughly assessed by the Therapeutic Goods Administration (TGA) and found to be effective. Read more about the approval of the vaccines.
Clinical trials of both vaccines also indicate their effectiveness is very good.
As other countries around the world progress with their rollouts, more and more data is becoming available that shows these vaccines are both highly effective when it comes to a real world situation.
For example, data from the United Kingdom shows that both these vaccines also significantly reduce severe COVID-19 in older adults.
This adds to the growing evidence that shows both the Pfizer (Comirnaty) and AstraZeneca (Vaxzevria) vaccines are working and are highly effective in protecting people against severe illness, hospitalisation and death.
All viruses mutate. COVID-19 is no different and there have been reports in media recently about new strains of the virus.
This does not mean the vaccines will not be effective on new variants.
Evidence tells us that the COVID-19 vaccines will still be effective against these new variants.
The Therapeutic Goods Administration (TGA) will also closely look at this as part of their approval and monitoring processes.
It may mean people need booster shots like tetanus and whooping cough or it may mean we need to be vaccinated again – like we are for the flu vaccine each year.
Researchers are still investigating this, but they do know the virus has not mutated enough to make current vaccines ineffective.
All the vaccines that are currently approved for use in Australia and other countries have demonstrated they are highly effective in preventing severe illness from COVID-19.
Click here to book an appointment at M3 Health Clinics
In the spirit of reconciliation M3 Health acknowledges the traditional custodians of country throughout Australia and their connections to land, sea and community. We pay our respect to their elders, past and present and extend that respect to all aboriginal and Torres strait islander peoples today.
Copyright © 2026 M3 Health